Cyclophosphamide
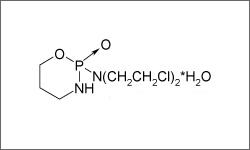
Cyclophosphamide (Cytoxan®, Neosar®, Cytoxan®IV, Procytox®) is an alkylating agent used singly or as part of a combination to treat a wide variety of neoplastic diseases. Malignancies for which cyclophosphamide is used include: Burkitt's lymphoma, Bladder cancer, Bone cancer, Cervical cancer, Endometrial cancer, Lung cancer and Prostate cancer, Testicular cancer and Cancer of the adrenal cortex. Cyclophosphamide may be administered orally, in the form of an oral solution or tablets, or through injections.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Cyclophosphamide is activated by liver cytochromes, which form cross-links with DNA and results in inhibition of DNA synthesis and function.1
The molecular structure below shows the 3 D conformer Cyclophosphamide.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Common side effects include: hair loss (alopecia), nausea and vomiting, diarrhea, depression of blood cell counts, loss of appetite, irritation of the bladder, cough, fever and/or chills, lower back or side pain, as well as skin and mouth ulcers. Less common side effects and toxicities include effects on the germ cells resulting in amenorrhea (cessation of menstrual periods) testicular atrophy, and sterility. For these reasons patients considering cyclophosphamide therapy should talk to their doctor about the drug's effects on fertility, potential or current pregnancy, and breast-feeding. Secondary cancers may appear years after cyclphosphamide therapy. Before beginning treatment with cyclophosphamide patients should discuss all other current medications with their physician.1
- 1 Cytoxan.. Prescribing Information. Bristol-Myers Squibb. 2016. [http://www.bms.com]
