Fulvestrant
Fulvestrant is used to treat postmenopausal women with estrogen receptor positive metastatic breast cancer that has progressed after treatment with antiestrogen therapy. Fulvestrant is given as an injection once a month.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Fulvestrant (Faslodex®) is an estrogen receptor antagonist. It binds reversibly to estrogen receptors instead of estrogen. Therefore, even though estrogen is still made, it is unable to trigger cancer cell growth because the receptors are blocked. The drug also leads to a decrease in the concentration of receptors present in cells.1
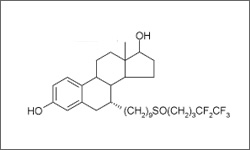
The structure above shows the 3D molecular structure of Fulvestrant.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Common side effects include nausea, vomiting, constipation, diarrhea, abdominal pain, headache, back pain, hot flashes, sore throat. Patients should not become pregnant while being treated with Fulvestrant as it may cause harm to the unborn child.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
