Vorinostat
Vorinostat is approved to treat cutaneous manifestations in patients with cutaneous T-cell lymphoma (CTCL) that is persistent or recurrent after two prior treatments. Treatment with vorinostat is a once daily oral capsule which is generally taken with food.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Vorinostat (Zolinza®) is a histone deacetylase (HDAC) inhibitor. The anti-cancer mechanisms of vorinostat are not fully known, but it is believed that the HDAC inhibition opens chromatin to allow for gene transcription and inhibition of cell division. In cancer cells, HDACs are often overexpressed, creating a condensed chromatin structure that inhibits gene transcription, and in turn can lead to apoptosis.1
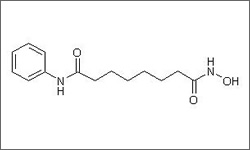
The diagram below shows the 3D molecular structure of Vorinostat.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Side Effects include: Low platelet and red blood cell counts, Nausea, vomiting, anorexia, constipation and diarrhea, Hyperglycemia, Fatigue and chills, Taste disorders.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Patients receiving anticoagulants should be closely monitored during vorinostat treatment due to the potential for increased prothrombin time and international normalized ratio (INR). Pulmonary embolism and deep vein thrombosis have also been observed in some cases. Use of vorinostat along with other HDAC inhibitors can produce decreased platelet counts and gastrointestinal bleeding. Women should avoid becoming pregnant during vorinostat treatment due to the possibility of developmental defects in the fetus. Women who are already pregnant should be warned of this risk. New mothers who decide to begin treatment with vorinostat should discontinue nursing. Since vorinostat is eliminated via metabolism by the liver, patients with liver failure should be treated with caution.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
