In addition to chemicals and radiation, another source of mutation is viruses. Viruses are very small 'organisms' that can infect the cells of other animals or plants. Humans are susceptible to a large number of different viruses. Viruses are not the same as bacteria although both can cause human disease. Treatments that cure bacterial infections are not useful in the treatment of viral infection. Some examples of viruses include the agent that causes the flu (influenza virus) and the causative agent of AIDS, the human immunodeficiency virus (HIV).
Viruses can disrupt cell behavior in several different ways.
- They can directly cause DNA damage (mutations) by inserting their genomes into the DNA of the host cell. The integration can disrupt important regulatory genes.
- The viruses may contain their own genes that disrupt the regulation of the cell. This process may be beneficial to the virus if it allows for rapid production of progeny but can be seriously detrimental to the host.
- Some viruses actually carry altered versions of genes that they have picked up from previous host cells. These altered genes no longer function properly, and when they are inserted into a new host cell, they cause disregulation and can lead to cancerous growth.
Through their mutagenic activity or their effects on cell behavior, viruses play a significant role in the development of particular cancers in many different animals, including humans.
Viruses have also been a major target of scientific investigation with respect to cancer. Some of the earliest work on the identification of oncogenes and tumor suppressors utilized viruses.1
Viruses can be divided into two rough categories, those that have DNA as their genetic material and those that have RNA as their genetic material. Both kinds of virus have been found to be associated with cancers of different types. Further information on the topics on this page can also be found in most introductory Biology textbooks, we recommend Campbell Biology, 11th edition.2 The viruses known to cause human cancers are:
- Epstein-Barr Virus (EBV) - Burkitt's Lymphoma
- Hepatitis B Virus (HBV) - Liver Cancer
- Hepatitis C Virus (HCV) - Liver Cancer
- Human Herpesvirus 8 (HH8) - Kaposi's Sarcoma
- Human Papillomavirus (HPV) - Cervical Cancer, Head and Neck Cancers, Anal, Oral, Pharyngeal, and Penile Cancers
- Human T-cell Lymphotropic Virus 1 (HTLV) - Adult T-cell Leukemia
- Merkel Cell Polyomavirus - Skin Cancer (Merkel Cell Carcinoma)
Epstein-Barr Virus (EBV) - Burkitt's lymphoma
Associated Cancer: Lymphoproliferative disease, most commonly Burkitt's Lymphoma. There is increasing evidence EBV is also associated with Hodgkin lymphoma.
Prevalence: It's estimated that more than 90% of the World population is infected with EBV. EBV is responsible for infectious mononucleosis (the 'kissing disease')
Transmission: Mechanism of transmission generally unknown, possibly through saliva.
Infection: EBV Infection usually begins in the epithelial cells of the oropharynx, posterior nasopharynx and parathyroid glands. From there EBV infects B cells and persistent infection is established. Almost all cases of EBV infection are controlled by the immune system and infected individuals are asymptomatic (have no symptoms of infection).
Carcinogenic Potential: B cell Infection is necessary for EBV mediated carcinogenesis. Only a small percentage of infections lead to cancer, most cases arising in immunocompromised or transplanted individuals. These patients are especially susceptible because they lack sufficient immune function to inhibit the growth of infected B cells. EBV-mediated carcinogenesis is most likely caused by the actions of viral gene products. Two proteins in particular are thought to play a major role in B cell immortalization; latent membrane proteins (LMP's) and EBV nuclear antigen (EBNA's). LMP1 is inserted into the host cell membrane and acts as an activated growth factor receptor, resulting in unregulated growth. EBNA's affect the cell in many different ways; one pathway leads to altered activity of tumor suppressors including Rb, p53, and Arf.
Read more about Burkitt's Lymphoma
Hepatitis B Virus (HBV) - Liver Cancer3, 5
Associated Cancer: Human Hepatocellular Carcinoma (HCC)
Prevalence: HBV is prevalent on every continent, but is especially high in sub-Saharan Africa and Southeast Asia. It is estimated that over 400 million people worldwide are infected.
Transmission: HBV is transmitted via contact with contaminated blood, sweat, or tears. It can also be spread through sexual contact and from mother to child.
Infection: HBV Infection occurs mainly in the liver, but viral antigens can be detected in the blood throughout the body. Chronic Infection is indicated by the presence of viral antigens in the blood for longer than 6 months. Chronic Infection can lead to cirrhosis of the liver and development of HCC.
Carcinogenic Potential: Hepatitis viruses (B and C) are responsible for 70-85% of primary liver cancers. Viral integration into the host genome is regularly found in chronic liver infection and cancer. This suggests it plays an important role in carcinogenesis. HBV encodes a protein (HBX) that may promote cell proliferation and interfere with DNA repair. HBV mediated carcinogenesis is likely due to a number of factors; the oncogenic properties of HBX, chronic liver damage, chronic inflammation, and the continuous tissue regeneration needed to maintain the liver during an infection.
Hepatitis C Virus (HCV) - Liver Cancer3, 6
Associated Cancer: Human Hepatocellular Carcinoma (HCC)
Prevalence: HCV is prevalent on every continent, but it is especially high in sub-Saharan Africa and Southeast Asia. It's estimated over 170 million people are infected with HCV worldwide.
Transmission: HCV is transmitted primarily through blood-blood contact.
Infection: HCV Infection occurs mainly in the liver and produces a more severe inflammation than HBV. Over 80% of those infected with HCV will develop cirrhosis of the liver or HCC.
Carcinogenic Potential: Hepatitis B and C are responsible for 70-85% of primary liver cancers. HCV is not known to integrate into the host genome. In vitro HCV has been shown to affect a wide range of cellular mechanisms that may promote carcinogenesis, but these have not been reproduced in vivo. The carcinogenic potential of HCV lies in its ability to cause chronic immune response-mediated hepatic damage, inflammation, and tissue regeneration.
Human Herpesvirus 8 (HHV8) - Kaposi's Sarcoma
Human Herpesvirus 8, aka Kaposi's Sarcoma Herpesvirus (KSHV) 3, 7
Associated Cancer: HHV8 primarily causes Kaposi's Sarcoma (KS), a type of cancer that affects the skin and soft organs. HHV8 is also associated with several blood disorders.
Prevalence: HHV8 is uncommon in most of the world, only 1-5% of people in North America and Northern Europe are infected. Mediterranean populations have a higher infection rate (5-20%) and Sub-Saharan Africa has the highest rate (>60%). In the U.S., gay men also have a higher infection rate (~40%).
Transmission: HHV8 is most commonly spread through sexual contact and via saliva. Transmission also may occur via organ transplantation or blood transfusion.
Infection: HHV8 infects B cells, epithelial cells, endothelial cells and possibly monocytes. HHV8 infection is high in populations with high incidence of KS and low in populations with low incidence of KS.
Carcinogenic Potential: HHV8 DNA is found in all cases of KS, but infection is not enough to cause cancer. The exact method by which HH8V induces cancer is still under investigation. KS probably starts as an inflammatory process to which circulating cells (including HHV8 infected cells) are recruited, leading to further inflammation, tissue damage, and viral infection. HHV8 then establishes a persistent infection which may send signals promoting angiogenesis and inflammation. This cycle may ultimately lead to tumor development. Untreated AIDS confers a 20,000 fold higher risk of developing KS, but other than immunosuppression the role of AIDS is generally unknown.
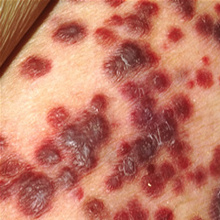
Kaposi's Sarcoma - Image Courtesy NCI
Human Papillomavirus (HPV) - Cervical Cancer, Head and Neck Cancer, Anal, Oral, Pharyngeal, and Penile Cancers
What is the Human Papillomavirus (HPV)?
There are over 200 known genetically different strains of human papillomavirus. Some strains of the virus are responsible for common warts and plantar warts (warts on the bottom of the feet). Other strains can infect the inner lining of the cervix; these strains are divided into low risk and high risk strains. The high risk strains are those strongly associated with cervical cancer. Two high risk strains, 16 and 18, are thought to be responsible for over 70% of cervical cancer cases. Low risk strains 6 and 11 are responsible for a large percentage of genital warts.8
Other than the appearance of genital warts, infection with the human papillomavirus is asymptomatic (doesn't have symptoms). In most cases HPV doesn't cause any problems and is cleared by the immune system.9
Prevalence
A 2007 study in United States found approximately 26.8% of all women examined (aged 14-59) were infected with the human papillomavirus. It is important to note that most HPV infections are cleared within two years (i.e. most infections are not lifelong).10
- 14-19 ~ 24.5%
- 20-24 ~ 44.8%
- 25-29 ~ 27.4%
- 30-39 ~ 27.5%
- 40-49 ~ 25.2%
- 50-59 ~ 19.6%
Transmission
The human papillomavirus is transmitted via skin-skin contact. Sexual intercourse is not necessary for transmission, but is the most common route. The virus can infect the genital, anal, and oral regions of the body. Infection occurs when viruses enter into small breaks in the skin or mucous membranes. The probability of acquiring HPV from a single sexual encounter is not known, but is probably high.11
HPV and Genital Warts
Some types of HPV can cause warts on or around the genitals or the anus. The two types that cause most cases of genital warts are 6 and 11. These are low-risk strains and are not associated with cervical cancer.
HPV and Cervical Cancer
The human papillomavirus is the primary risk factor for cervical cancer. It is responsible for at least 90% of cases. High risk HPV type 16 is the most common high-risk type and is found in over 50% of cervical cancers. The second most common high-risk type is 18 and it is responsible for 10-12% of cervical cancers. Although HPV responsible for virtually all cases of cervical cancer, 80% of women clear an infection within 1-2 years.
There are 3 steps necessary for cervical cancer development:12
- HPV Infection. HPV infection is quite common; a recent study estimates over 26% of women in the United States are infected.
- HPV Persistence. Less common. In most women (~80%), HPV infections are cleared or suppressed by the immune system within 1-2 years of exposure. Therefore, most cases of HPV infection do NOT cause cervical cancer. If HPV infection is not cleared and persists for a long period of time a condition called cervical intraepithelial neoplasia (CIN) can develop. See below for more about cervical intraepithelial neoplasia.
- Cell Transformation and Invasion. Rare in comparison to the number of women infected by HPV. Invasion usually occurs after a long period of infection. It is thought that severe dysplasia does not occur without certain co-factors. (e.g. Smoking, HIV co-infection, Immunosuppression)
HPV and Other Types of Cancer
It is well known that HPV is a direct cause of cervical cancer, but its role in cancer may be much larger. Recently, studies have strongly linked HPV with cancers of the vulva, penis, anal canal, and head-and-neck.13
- 73% of oral cavity tumors tested positive for HPV-16 DNA.14
- 80% of anal cancers positive for either HPV-16 or 18 DNA.
In 2012, a population (epidemiologic) study showed an association between HPV subtypes that infect skin (called cutaneous infections) and the development of squamous cell skin cancer (SCC). The role of the virus in the development of the disease is not yet clear.15
How HPV Causes Cancer
HPV invades the skin or mucosa by entering tiny breaks in the surface (even those not visible to the naked eye). Once inside, HPV infects host epithelial cells, tricking them into producing new viruses. In the process of normal cell replacement, the infected cells are shed, releasing viral particles. High risk strains of HPV can integrate viral DNA into the host genome, although this is not a normal part of the HPV life cycle. Viral integration may give infected host cells a selective advantage, leading to a longer infection time. The longer the infection lasts, the more time there is for cancer to develop.16
After integration, two viral genes (E6 and E7) may be over-expressed. The E6 and E7 proteins are responsible for the ability of HPV to cause cancer. The E6 and E7 proteins prevent the activity of key tumor suppressors. E6 inhibits p53, a protein that controls responses to different types of cellular stress including DNA damage and viral infection. E7 inhibits Rb, a protein that can prevent cell division by blocking the activity of transcription factors. The combined effects of E6 and E7 put cells at risk for undergoing uncontrolled division that can lead to cancer.17, 18
The HPV genome contains several genes that encode proteins.[1] In HPV, three of these genes, E2, E6, and E7, are of particular interest because of their roles in the development of cervical cancer. The E2 protein functions by binding to both the E6 and E7 proteins. When E6 and E7 are bound to E2 they are blocked from acting in the cell. [2] When E6 is not bound to E2, it is free to bind to the p53 tumor suppressor. When E6 binds to p53, p53 is destroyed and cannot function. p53 is a key protein in cell cycle control. It is not functional in over 50% of all human cancers. Without the p53 protein, a cell may continue to divide even if it is damaged.[3] The E6 protein also causes the expression of telomerase. [4] Telomerase is a protein that is not normally produced by most cells in adult humans. When it is present, telomerase maintains the ends of the chromosomes. This prevents the breakdown of chromosomes and helps cancer cells to to divide forever. [5]
Another HPV protein, E7, also plays a key role in helping the virus take over control of infected cells. When not bound by E2, E7 binds to another protein, Rb. When E7 is bound to Rb, Rb cannot carry out its normal function. Normally, Rb binds to E2F. E2F is a transcription factor that causes cell cycle progression. When E2F is bound to Rb, it cannot act as a transcription factor and cannot cause the cell to divide. But, if E7 binds to Rb, E2F cannot also bind to Rb and is then free to act as a transcription factor. In essence, E7 inhibits an inhibitor or cell division. When a cell makes the E7 protein, the E2F transcription factor causes the cell to divide, a critical step in cancer development. [6] The E6 and E7 proteins help HPV hijack cell division and help drive cancer development.
Could HPV Help Prevent Cancer?
Human papillomaviruses cause cancer, but they may also PROTECT against cancer. Research in mice showed that infection with a group of human papillomaviruses (beta-HPV) led to a protective immune response against squamous cell skin cancer (SCC). The researchers believe that the immune response against the virus somehow protects against cancers caused by exposure to ultraviolet (UV) radiation. They are now looking into using HPV vaccination to protect against skin cancer.19
Human T-cell Lymphotropic Virus 1 (HTLV) - Adult T-cell Leukemia
Human T-cell Lymphotropic Virus 1 (HTLV-1)3, 20
Associated Cancer: Adult T-cell Leukemia (ATLL)
Prevalence: Approximately 10 to 20 million people are infected worldwide. HTLV-1 is endemic in southwest Japan, in and around the Caribbean islands, in parts of Central Africa and South America.
Transmission: HTLV1 can be transmitted via sexual or blood-blood contact. It can also be passed through breast milk and from mother to fetus.
Infection: HTLV-1 is an RNA virus (a retrovirus) that can infect T-cells, B-lymphocytes, monocytes, and fibroblasts.
Carcinogenic Potential: About 3-5% of people with HTLV1 will develop adult T-cell leukemia, with most cases occurring in middle aged people. At least one of HTLV1's regulatory proteins, (Tax), is thought to be involved with ATLL development. Tax may contribute to carcinogenesis by inducing cellular proliferation, activating cell survival proteins, and also may contribute to chromosomal instability.21, 22
Several other retroviruses are also associated with cancer and the use of retroviral vectors in cancer treatment has to take into consideration the possibility that the treatments could cause major problems.23, 24
Merkel Cell Polyomavirus - Skin Cancer (Merkel Cell Carcinoma)
Merkel cells are located in the outer layer of skin (epidermis) and their exact function is not known. They appear to have sensory roles (i.e. detecting textures) and may have other functions.25, 26
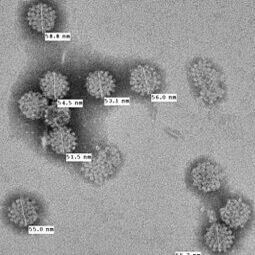
Merkel cell polyomavirus (MCV or MCPyV) has been linked to a form of skin cancer that affects Merkel cells - Merkel cell carcinoma.27 Whether the virus causes the cancer is not entirely clear. Researchers are also looking to see if MCV is associated with other types of skin cancer (squamous cell carcinoma - SCC and basal cell carcinoma - BCC).28, 29
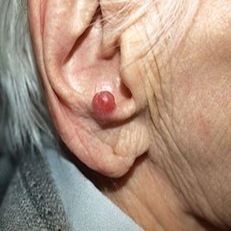
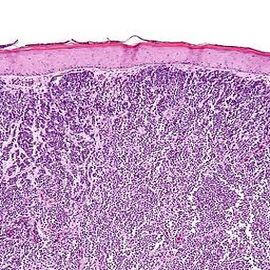
The images below, in clockwise order - a Merkel cell tumor, Merkel cell cancer as viewed under a microscope, virus-like particles from MCV, .
- 1 Cooper G. Oncogenes. Jones and Bartlett Publishers, 1995. 151-152, 175-176.
- 2 Urry, L. A., Cain, M. L., Wasserman, S. A., Minorsky, P. V., & Reece, J. B. (2017). Campbell Biology (11th ed.). Pearson.
- 3abcde E. Boccardo and LL Villa. Viral Origins of Human Cancer. Current Medicinal Chemistry. 2007; 24: 2526-39. [PUBMED]
- 4 Brady G, Macarthur GJ, Farrell PJ. Epstein-Barr virus and Burkitt lymphoma. Postgrad Med J. 2008 Jul;84(993):372-7. [PUBMED]
- 5 Lupberger J, Hildt E. Hepatitis B virus-induced oncogenesis. World J Gastroenterol. 2007 Jan 7;13(1):74-81. [PUBMED]
- 6 Castello G, Scala S, Palmieri G, Curley SA, Izzo F. HCV-related hepatocellular carcinoma: From chronic inflammation to cancer. Clin Immunol. 2010 Mar;134(3):237-50. Epub 2009 Nov 11. [PUBMED]
- 7 B. Ensoli, C. Sgadari, et al. Biology of Kaposi's sarcoma. European Journal of Cancer. 2001; 37: 1251-1269. [PUBMED]
- 8 Schiffman M, Castle PE, Jeronimo J, Rodriguez AC, Wacholder S. "Human papillomavirus and cervical cancer." Lancet. 2007 Sep 8;370(9590):890-907. Review. [PUBMED]
- 9 zur Hausen H. Papillomaviruses and cancer: from basic studies to clinical application. Nature Reviews. Cancer. 2002; 2(5):342-50 [PUBMED]
- 10 Dunne EF, Unger ER, Sternberg M, McQuillan G, Swan DC, Patel SS, Markowitz LE. Prevalence of HPV infection among females in the United States. JAMA. 2007;297(8):813-9. [PUBMED]
- 11 Burchell AN, Winer RL, de Sanjosé S, Franco EL. Chapter 6: Epidemiology and transmission dynamics of genital HPV infection. Vaccine. 2006 Aug 31;24 Suppl 3:S3/52-61. Epub 2006 Jun 2. [PUBMED]
- 12 Schiffman M, Castle PE. Human papillomavirus: epidemiology and public health. Archives of Pathology and Laboratory Medicine. 2003; 127(8):930-4. [PUBMED]
- 13 Chaturvedi AK. Beyond cervical cancer: burden of other HPV-related cancers among men and women. J Adolesc Health. 2010 Apr;46(4 Suppl):S20-6. [PUBMED]
- 14 D'Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, Westra WH, Gillison ML. Case-control study of human papillomavirus and oropharyngeal cancer. New England Journal of Medicine. 2007;356(19):1944-56. [PUBMED]
- 15 Iannacone MR, Gheit T, Waterboer T, Giuliano AR, Messina JL, Fenske NA, Cherpelis BS, Sondak VK, Roetzheim RG, Michael KM, Tommasino M, Pawlita M, Rollison DE. Case-control study of cutaneous human papillomaviruses in squamous cell carcinomas of the skin. Cancer Epidemiol Biomarkers Prev. 2012 Jun 15. [Epub ahead of print] [PUBMED]
- 16 Hamid NA, Brown C, Gaston K. The regulation of cell proliferation by the papillomavirus early proteins. Cell Mol Life Sci. 2009 May;66(10):1700-17. [PUBMED]
- 17 Jabbar S, Strati K, Shin MK, Pitot HC, Lambert PF. Human papillomavirus type 16 E6 and E7 oncoproteins act synergistically to cause head and neck cancer in mice. Virology. 2010 Aug 24. [Epub ahead of print] [PUBMED]
- 18 Moody CA, Laimins LA. Human papillomavirus oncoproteins: pathways to transformation. Nat Rev Cancer. 2010 Aug;10(8):550-60. Epub 2010 Jul 1. [PUBMED]
- 19 Strickley JD, Messerschmidt JL, Awad ME, et al. Immunity to commensal papillomaviruses protects against skin cancer. Nature. 2019;575(7783):519–522. doi:10.1038/s41586-019-1719-9[PUBMED]
- 20 Masao Matsuoka and Kuan-Teh Jeang. Human T-Cell Leukemia Virus Type I at Age 25: A Progress Report. Cancer Research. 2005; 65(11):4467-70. [PUBMED]
- 21 Boxus M, Willems L. Mechanisms of HTLV-1 persistence and transformation. Br J Cancer. 2009 Nov 3;101(9):1497-501. Epub 2009 Sep 29. [PUBMED]
- 22 Chlichlia K, Khazaie K. HTLV-1 Tax: Linking transformation, DNA damage and apoptotic T-cell death. Chem Biol Interact. 2010 Nov 5;188(2):359-65. Epub 2010 Jun 15. [PUBMED]
- 23 Nair V. Retrovirus-induced oncogenesis and safety of retroviral vectors. Curr Opin Mol Ther. 2008 Oct;10(5):431-8. [PUBMED]
- 24 Ruprecht K, Mayer J, Sauter M, Roemer K, Mueller-Lantzsch N. Endogenous retroviruses and cancer. Cell Mol Life Sci. 2008 Nov;65(21):3366-82. [PUBMED]
- 25 Moll I, Roessler M, Brandner JM, Eispert AC, Houdek P, Moll R. Human Merkel cells--aspects of cell biology, distribution and functions. Eur J Cell Biol. 2005 Mar;84(2-3):259-71. [PUBMED]
- 26 Maricich SM, Morrison KM, Mathes EL, Brewer BM. Rodents rely on Merkel cells for texture discrimination tasks. J Neurosci. 2012 Mar 7;32(10):3296-300. [PUBMED]
- 27 Chang Y, Moore PS. Merkel cell carcinoma: a virus-induced human cancer. Annu Rev Pathol. 2012 7:123-44. Epub 2011 Sep 13. [PUBMED]
- 28 Ganzenmueller T, Yakushko Y, Kluba J, Henke-Gendo C, Gutzmer R, Schulz TF. Next-generation sequencing fails to identify human virus sequences in cutaneous squamous cell carcinoma Int J Cancer. 2012 Apr 5. doi: 10.1002/ijc.27581. [Epub ahead of print] [PUBMED]
- 29 Rollison DE, Giuliano AR, Messina JL, Fenske NA, Cherpelis BS, Sondak VK, Roetzheim RG, Iannacone MR, Michael KM, Gheit T, Waterboer T, Tommasino M, Pawlita M. Case-control study of Merkel cell polyomavirus infection and cutaneous squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 2012 Jan;21(1):74-81. Epub 2011 Oct 20. [PUBMED]
