All cells respond to signals sent from other cells. One main way that cells communicate is hormones. Hormones can be either small proteins (like insulin) or they can be derived from steroids (like estrogen and testosterone). The signaling cell produces the hormone that floats over and binds to (or enters) the target cell to cause a change in behavior. Hormones can cause a cell to produce new proteins or to stop making products that were already being produced. One possible outcome of hormone signals is the growth and reproduction of the target cells.
Cancer cells, while abnormal, may retain the ability to respond to hormonal signals. The idea behind the majority of hormone-based cancer treatments is to starve the cancer cells of the hormonal signals that would otherwise stimulate them to divide. The drugs used in these treatments work by blocking the activity of the hormone in the target cell. Some newer treatments under investigation are designed to prevent the production of the hormone, cutting off the signal at the start. Some forms of breast, ovarian and prostate cancer are subject to hormonal treatments.
Hormonal treatments are often combined with surgery and/or radiation and/or chemotherapy. In these situations, the hormonal treatments are referred to as an 'adjuvant' treatment.

Hormones are signals made in one cell that affect different cells. In the animation above, the green chemical is turned into the red hormone by a protein machine (enzyme). Some hormones can cause cancer cells to reproduce.
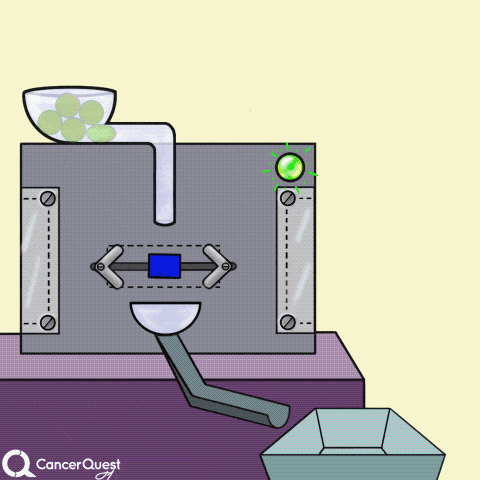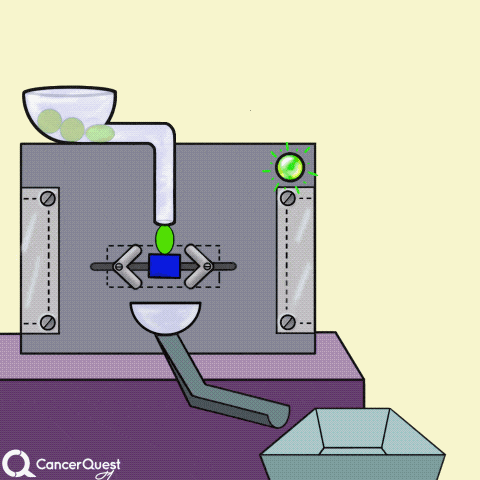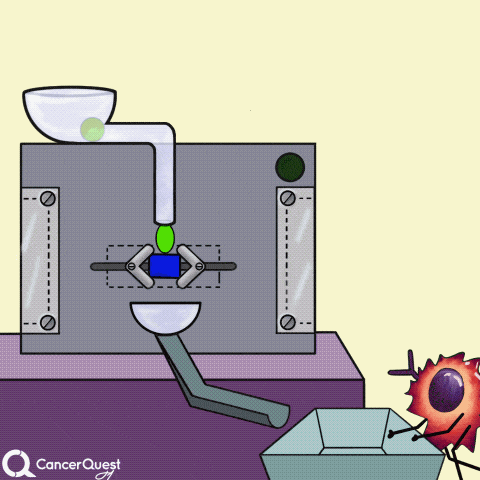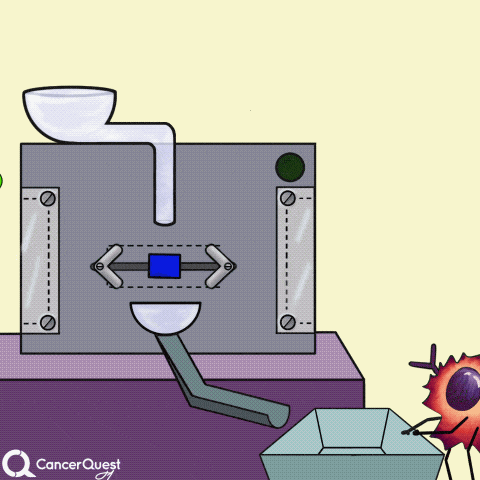
Some cancer drugs (blue rectangle) block the enzymes that produce hormones (like estrogen).

Cancer drugs (blue triangle) can also prevent hormones from sticking to or entering cancer cells. They can also 'jam' hormone receptors inside cells. Either way, the hormone's signal is blocked.
The hormonal treatments described below all work by interfering with hormonal signals but, as shown above, they may attack different parts of the signaling pathways to slow cancer growth. The different kinds of hormone inhibitors (also called 'antagonists') include:
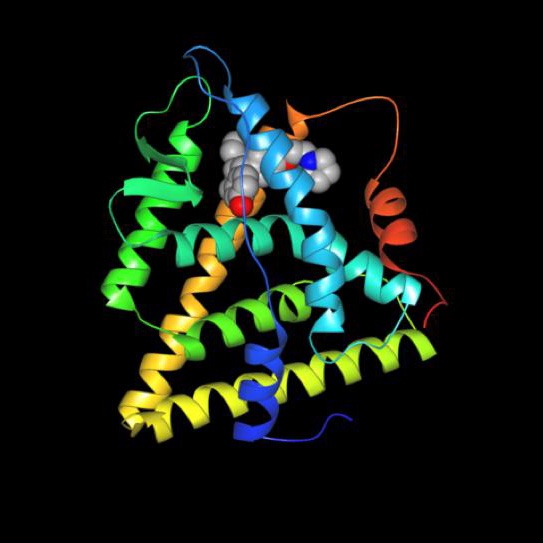
- Selective Estrogen Receptor Modulators (SERMs): These agents work by interfering with the activity of the estrogen receptor. Drugs that work this way include tamoxifen (Nolvadex®), raloxifene (Evista®) and toremifene (Fareston®)
- Aromatase Inhibitors: These drugs work by blocking the production of estrogen by the enzyme that makes it from its precursor. Drugs that work this way include anastrozole (Arimidex®), exemestane (Aromasin®) and letrozole (Femara®).
- Receptor Down-regulators: This class of drugs works by lowering the levels of the receptor for estrogen. Fulvestrant (Faslodex®) is a drug in this category.
- Selective Androgen Receptor Modulators (SARMs): These agents work by interfering with the activity of the androgen receptor. Drugs that work this way include flutamide (Eulexin®) and bicalutamide (Casodex®).
- Other Hormone Treatments: These drugs work to affect the production or activity of hormones in a manner that is not covered in the above categories. The way by which these drugs work may not be entirely clear. An example of this type of drug is megastrol (Megace®).
- Glossary of Hormonal Treatment: An easy to use table of hormonal treatment drugs including trade name, generic name, and type. Contains links to additional information.
- Know the Flow: Hormonal Treatments: An interactive self-test about hormonal treatments for cancer.
Selective Estrogen Receptor Modulators (SERMs)
Many individuals with breast cancer have tumors driven to grow by the naturally occurring hormone, estrogen . One of estrogen's normal activities is to cause the proliferation of cells in the breast and uterus; each month new cell linings must be created for the milk glands (breast) and the endometrium (uterus). In some breast cancer patients this normal expression of estrogen contributes to the growth and division of the cancer cells. For these patients, treatment with selective estrogen receptor modulators (SERMs), also known as anti-estrogens, is appropriate.
The drugs work by causing changes in the shape of estrogen receptors (ER), preventing the action of the hormone. The drugs only affect a subset of the cells capable of responding to estrogen but the precise mechanism of this selectivity is not yet known. It is this selective action that has earned the drugs their name. The blockage of estrogen in the target cells causes changes in gene expression and alters the behavior of the cells, preventing cell division.1
In 1992, tamoxifen became the first SERM to be used for the treatment of breast cancer. While it does decrease estrogenic effects in the breast, it unfortunately has a pro-estrogenic activity in the uterus, causing a rise in uterine cancer for tamoxifen-treated breast cancer patients.2
Recently, next generation SERMs such as raloxifene have been investigated for their potential as breast cancer treatments. This drug appears to have anti-estrogenic effects in both breast and uterine tissues. Two six-month studies conducted in 1999 show that raloxifene does not stimulate endometrial growth. Studies of 969 post-menopausal women taking raloxifene showed that treatment with the drug did not lead to a significant increase in endometrial thickness as compared to women taking a placebo.3
The results of the STAR trial showed that raloxifene was equal to tamoxifen for the prevention of invasive breast cancer in post-menopausal women and had somewhat improved side effects.4, 5, 6
In 2007, the U.S. Food and Drug Administration approved raloxifene hydrochloride tablets (EVISTA, Eli Lilly and Company) to reduce the risk of invasive breast cancer in postmenopausal women with osteoporosis and in postmenopausal women at high risk for invasive breast cancer.7, 8, 9
Watch an animation showing how these drugs work.
Tamoxifen (Nolvadex®)
Raloxifene (Evista®)
Toremifene (Fareston)
Inhibitors and Down Regulators
Additional hormonal treatments have been developed which can act as alternatives to treatment with SERMs (tamoxifen/raloxifene) or be utilized if SERMs are ineffective. Tamoxifen and raloxifene are both designed to interfere with the action of estrogen once it has entered a target cell. There are several other points of attack that may be taken to prevent signalling via estrogen. The most direct approach is to prevent the production of the estrogen by blocking the enzyme that is responsible for its formation, aromatase.
A second approach is to remove the target of the estrogen (the estrogen receptor). Both of these approaches to therapy have been developed for the treatment of breast cancer. Because these hormone treatments block estrogen production or function, they can have negative side effects on pregnancy. Therefore, they are used to treat postmenopausal women.10
Watch animations of the estrogen receptor and the action of the drug tamoxifen.
Aromatase Inhibitors
After menopause, women produce a consistent low level of estrogen that is derived from androgen precursors. These precursors are converted to estrogen through the actions of the enzyme aromatase. By blocking the action of this enzyme, aromatase inhibitors prevent the formation of estrogen. There are two types of aromatase inhibitors that have been approved as treatment for postmenopausal women with estrogen-receptor positive metastatic breast cancer: steroidal inhibitors such as exemestane (Aromasin®) and non-steriodal inhibitors that competitively bind to aromatase (anastrozole and letrozole).10
Anastrozole
Exemestane
Letrozole
An advantage of these drugs is that they do not apear to have the side effect of increasing the risk of endometrial cancer.10
Because they interfere with that activity of the sex hormone estrogen, aromatase inhibitors do have additional side effect of impacting sexual function and satisfaction.11
Estrogen Receptor Down-regulators
By decreasing the concentration of estrogen receptors and their activity, the effects of estrogen can be negated.
Selective Androgen Receptor Modulators (SARMs)
Testosterone and dihydroxytestosterone (DHT) are two hormones (androgens) produced in the testes and adrenal glands. Among other activities, these hormones bind to specific receptors in the cells of the prostate. A normal function of this binding is to regulate the growth of the prostate cells, however, in cancer cells this regulation is compromised. The androgens bind to the receptors in cancer cells and contribute to their growth and division.
Anti-androgen molecules have been created, that can enter cells and prevent the binding of testosterone to the receptor proteins. This occurs via the preferential binding of the drug molecule to the androgen receptors. The binding of the drug prevents the androgens from binding and therefore reduces their pro-growth activities.12
Bicalutamide(Casodex®)
Flutamide (Eulexin®)
Nilutamide (Nilandron®, Anandron®)
The drugs are frequently used in combination with surgery to cause the maximal reduction in androgen levels.13PUBMED] Additional drugs are also being studied for their ability to reduce androgen levels in prostate cancer patients and to overcome the drug resistance seen in some patients. 14, 15
Additional Hormone Treatments
Additional agents have been developed to interefere with the hormonal control of cell division. These agents may mimic naturally occuring hormones or may work through novel or unknown mechanisms. Examples of this type of drug include:
Megestrol (Megace): this drug interferes with the production and/or activity of estrogen. 16
Know the Flow: Hormonal Treatments
Know the Flow is an educational game for you to test your knowledge. To play:
- Drag the appropriate choices from the column on the right and place them in order in the boxes on the left. Note that you will only use five of the six choices to complete the game.
- When done, click on 'Check' to see how many you got correct.
- For incorrect answers, click on 'Description' to review information about the processes.
- To try again, choose 'Reset' and start over.
Please visit us on a larger screen to play this game.
- 1 Katzenellenbogen B, Katzenellenbogen J. "Defining the "S" in SERMs". Science (March 29, 2002). 295: 2380-2381. [PUBMED]
- 2 Shang, Y Brown, M. "Molecular Determinants for the Tissue Specificity of SERMS". Science (March 29, 2002). 295: 2465-2468. [PUBMED]
- 3 Scott J, Da Camara C, Early J. "Raloxifene:A Selective Esrogen Receptor Modulator". American Family Physician (September 1999). [http://www.aafp.org/afp/990915ap/1131.html]
- 4 Vogel, V., Costantino, J., Wickerham, L., Cronin, W., Cecchini, R., Atkins, J., … Project, N. (2006). Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial. JAMA, 295(23), 2727–41. (Original work published 2006年6月) [PUBMED]
- 5 Land SR; Wickerham DL; et.al. Patient-Reported Symptoms and Quality of Life During Treatment With Tamoxifen or Raloxifene for Breast Cancer Prevention: The NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;295:2742-2751. Published online June 5, 2006. [PUBMED]
- 6 Vogel VG The NSABP Study of Tamoxifen and Raloxifene (STAR) trial. Expert Rev Anticancer Ther. 2009 Jan;9(1):51-60. Erratum in: Expert Rev Anticancer Ther. 2009 Mar;9(3):388. [PUBMED]
- 7 https://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022042s000_Approv.pdf
- 8 https://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022042_evista_toc.cfm
- 9 "Understanding Estrogen Receptors, Tamoxifen and Raloxifene". National Cancer Institute. [http://newscenter.cancer.gov/sciencebehind/estrogen/estrogen00.htm]
- 10abc Bentram DJ. "Role of Antiestrogens and Aromatase Inhibitors in Breast Cancer Treatment." Current Opinion in Obstetrics and Gynecology (2002). 14: 5-12. [PUBMED]
- 11 Baumgart J, Nilsson K, Evers AS, Kallak TK, Poromaa IS. Sexual dysfunction in women on adjuvant endocrine therapy after breast cancer. Menopause. 2012 Sep 17. [Epub ahead of print] [PUBMED]
- 12 Nguyen MM, Wang Z. Manipulation of androgens and alterations in the androgen receptor axis in prostate cancer. Minerva Urol Nefrol. 2008 Mar;60(1):15-29. [PUBMED]
- 13 Klotz L. Maximal androgen blockade for advanced prostate cancer. Best Pract Res Clin Endocrinol Metab. 2008 Apr;22(2):331-40. [
- 14 Chen Y, Clegg NJ, Scher HI. Anti-androgens and androgen-depleting therapies in prostate cancer: new agents for an established target. Lancet Oncol. 2009 Oct;10(10):981-91. [PUBMED]
- 15 Klotz L. Maximal androgen blockade for advanced prostate cancer. Best Pract Res Clin Endocrinol Metab. 2008 Apr;22(2):331-40. [PUBMED]
- 16 Phase II study of high-dose megestrol acetate in platinum-refractory epithelial ovarian cancer. Wilailak, Sarikapan1; Linasmita, Vasant1; Srisupundit, Somkeart. Issue: Volume 12(9), October 2001, pp 719-724 [PUBMED]
